About Event
Transforming Your Flow Chemistry Economics with Scale-Up Efficiency, Safety & Success
Welcome to the 5th Commercializing Flow Chemistry Summit, returning this October to Boston, showcasing the latest strides in pharmaceutical manufacturing technologies. Since its inception, this summit has been instrumental in spearheading the integration of flow chemistry innovations within the industry, highlighting annual advancements in critical areas such as electrochemistry and continuous manufacturing. This year, the focus intensifies on tackling industry-wide challenges such as scalability and compliance, while unveiling new opportunities in digital twins and sustainable practices.
Don’t miss the chance to explore cutting-edge technologies and strategic insights essential for enhancing efficiency and sustainability in pharmaceutical manufacturing.
Join our CFC community & Explore
Optimizing Electrochemistry Integration for Enhanced Process Economics
Learn to implement electrochemical methods for cost-effective and scalable synthesis, enhancing production efficiencies with Merck
Navigating Strategic Flow Chemistry Adoption
Master a robust decision-making framework for choosing flow chemistry over traditional batch processes, focusing on efficiency, cost-effectiveness, and strategic adoption with Genentech
Strategizing Late-Stage API Process Development
Explore comprehensive strategies for transitioning API processes to continuous manufacturing, highlighting underappreciated opportunities and organizational alignment with AbbVie
Boosting Process Safety and Sustainability in Pharma
Explore specialty chemical practices that enhance safety protocols and environmental sustainability in flow chemistry, with a focus on adapting these methods to meet stringent regulatory standards with BASF
Optimizing Product Quality with PAT Tools
Discover how PAT enhances real-time monitoring and control of flow chemistry processes; understand reaction mechanisms and conditions to ensure product consistency and minimize waste with Takeda
Network Alongside
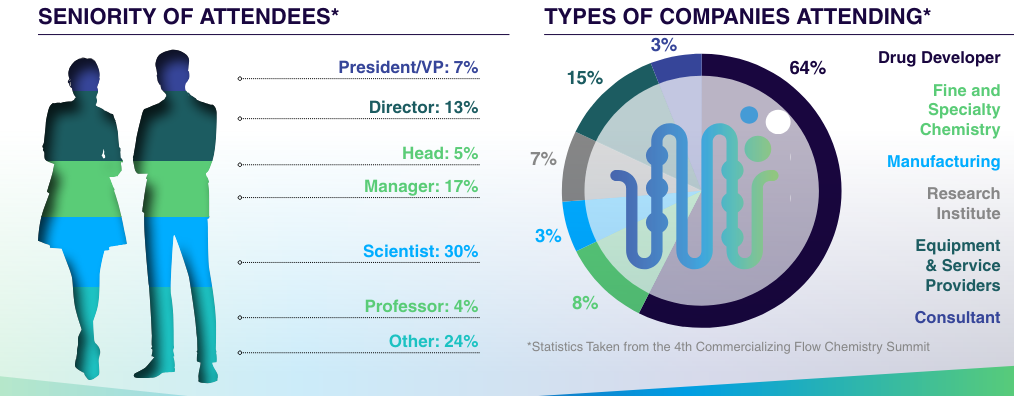
The 5th Commercializing Flow Chemistry Summit is tailored for senior professionals from biopharma, specialty, and fine chemical industries, including directors, VPs, principal scientists and engineers in process engineering and process chemistry. This event attracts these leaders due to its focus on cutting-edge process development, strategic discussion, and the latest innovations in continuous manufacturing, providing vital insights and networking opportunities to drive industry advancement.
What Your Peers Have to Say:
"Flow chemistry enhance the sustainability of chemical processes by reducing the waste and improving the efficiency, the 5th Commercializing Flow Chemistry Summit will likely address these aspects. I am looking forward to interact with the leading scientists and industry professional hoping for potential collaborations, partnerships and career opportunities"
Dr Firdaus Parveen, Liverpool Digital Flow Centre Manger and Research Coordinator, University of Liverpool and Material Innovation Factory, 2024 Speaker
"The talks had a good variety and were very engaging. Designated networking opportunities were great for connecting with other industry leaders and continuing thoughtful discussions. Roundtable discussions were useful for gaining a better perspective"
Nick Kaczorowski, Senior Engineer Process Development, Eli Lilly, 2023 Speaker
"Insightful talks, exciting topics and great networking event within the continuous flow field"
Thomas Toupy, Scientist, Massachusetts Institute of Technology, 2023 Speaker

